Ryan Plumadore, Wake Forest University School of Law JD ’26
The halls of St. Edwards Children’s Hospital are quiet in the mornings. Most of the movement comes from nurses changing shifts, and the slow and steady beeping of heart monitors drifting in from down the hall. Parents fidgeting in the cold, plastic seats of the waiting rooms, pensively awaiting the results of tests and treatments.
Room 513 is different from the rest of the hospital’s sterile halls. It is a quaint room at the end of the west hallway, a short walk from the nurses’ station and directly across from the window overlooking the courtyard garden. (In the springtime, beautiful flowers fill the courtyard. Right now, it’s mostly overgrown shrubs.) On the windowsills lay extra blankets neatly folded. One pink toothbrush sits on the bathroom sink. The table in the corner of the room overflows with watercolor paintings and sketches of superheroes like Wonder Woman. Some watercolor paintings hang on the entry door—faded with the passage of time. Strings of polaroid photos adorn the walls, each photo holding a snapshot of laughter and happy memories. Room 513 is Lila’s room—her familiar haven.
Lila sits up in her bed, finishing the last few bites of her toast. Lila eats slowly, not because she isn’t hungry but because she gets tired easily. Before all of this, Lila excelled on her high school track team. Now, she has to pace herself just to finish breakfast.
Lila pushes her breakfast tray aside and picks up her sketchbook where she flips to the next blank page. She draws the same way she eats: slowly. Bringing her pencil down to the page, she makes her first line: careful and steady, something she can control in the midst of her daily uncertainty.
Julie sits in the chair beside Lila’s bed; her hands folded loosely in her lap. She watches her daughter draw in quiet concentration. At first glance, it looks like any other morning—but Julie notices the little things: the soft tremor in Lila’s hand when she reaches for her pencil, the way she pauses between lines as if gathering the strength to take one breath at a time. Still, Lila pushes through her pain, her movement is steady and persistent, just like her hope to recover.
The room settles into the stillness that now feels familiar. The monitor beside the bed beeps with an irregular rhythm. Lila and Julie barely even notice it anymore. In the hallway, nurses laugh softly as they push supply carts down the hall. But inside Room 513, everything moves at Lila’s pace.
Julie rises from her chair and crosses the small room to the windowsill where the extra blankets are stacked. She reaches for the top one, but her hand slows as her eyes drift to the wall beside it—the one crowded with Lila’s paintings. A few of the paintings are bright and new, their colors still popping off the page. Most, though, were much older, made in the spring when Lila could finish a whole picture in one sitting. Julie feels a familiar lump rise in her throat. The room is a time capsule.
“You doing okay?” Julie asks in a soft tone.
Lila nods without looking up, her pencil moving in small, soft strokes. Speaking would cost her more energy than she can spare this morning, and Julie knows better than to ask. She’s learned to listen to the small cues, how Lila answers with a gesture instead of words, how her breathing shifts when she’s tired.
Julie returns to her chair and gently drapes the blanket over Lila’s feet. Lila keeps drawing, her attention fixed on the page as she patiently fills out another corner of her sketchbook, one careful line at a time.
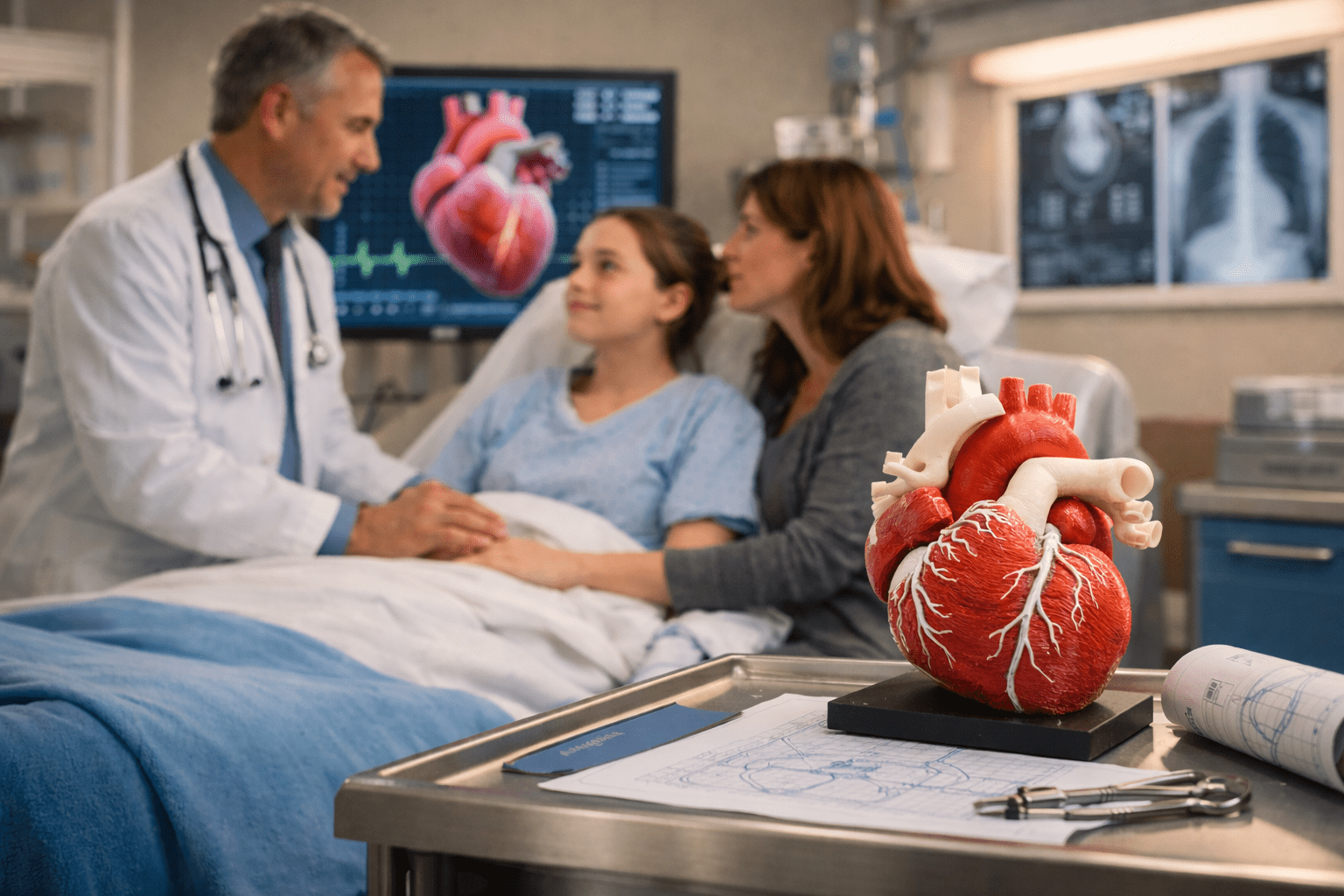
A soft knock at the door breaks the silence in the room. Dr. Jones steps into the doorway with a small, reassuring smile, his tablet tucked under one arm. “Good morning, Lila,” he says softly as he enters the room. “How are you doing in here?”
He closes the door behind him; the soft click breaks the silence in the room. Julie returns a tired smile. Dr. Jones approaches the bedside monitor, its soft light reflecting off the metal rails of the bed. Julie catches his hesitation instantly. She sits a little straighter, grounding herself before Lila has a chance to notice.
Dr. Jones pulls a chair to Lila’s bedside, its rubber wheels squeaking across the room. He balances his tablet on his knee as he begins a new set of morning notes.
“Lila,” he says gently, “I’m going to ask you a few things, alright? You can just nod.”
Lila gives a small nod, still filling out the corner of her sketchbook.
“Any dizziness this morning?”
Another nod.
“Shortness of breath before breakfast?”
Lila nods again, slower this time.
Julie clears her throat, “She needed a minute this morning before she could sit up.”
Dr. Jones uses a stylus to enter this into his notes. He sets it aside on the windowsill and shifts closer. Leaning in, he places the stethoscope against Lila’s chest. The sound is steady but uneven. Julie watches his expression as he listens.
When he finishes, Dr. Jones lets out a short breath and pulls his chair back to face them.
“Her vitals are holding, but we’re seeing the same patterns we saw last week. The new medications are keeping her stable, but they are not moving her forward.”
Julie’s gaze drops to her hands. “So are we running out of time?”
Dr. Jones shakes his head, “We’re running out of options,” he says. “The goal was to show improvement, but we have seen none in the last month.”
Lila’s pencil freezes mid-sketch.
Dr. Jones continues, “We need to start thinking about other options. Something that will actually relieve the pressure on her arteries rather than just managing her symptoms.”
Julie looks up, “A surgery?” She asks.
“There are less invasive procedures than an open surgery,” says Dr. Jones, “Still serious, but we need to find the right device to fit Lila.”
Julie’s eyes wander to the wall of watercolors— the bright blues, the faded yellows, and the capes of superheroes wa ving around mid-flight— and then back to him. “But I thought you said we don’t have that.”
“Not today,” he continues. “But there is something in development. A device that I have been following closely for months. It is designed especially for teenagers like Lila. Not a repurposed adult stent, but one that is matched to her anatomy.”
Julie’s voice tightens. “How close is it to being available?”
“Very close. It’s just waiting for final approval. The moment it’s cleared, I want us to be ready to make a decision.”
He rises, smoothing out the wrinkles on his sterile lab coat. Before leaving, he leans toward Lila with a soft smile. “I will check in on you again this afternoon. If anything changes, call right away.”
He reaches for the doorknob, but pauses and glances at Julie, his expression softens. “We’re not done fighting this.”
* * *
Two floors above, on the research wing of St. Edwards Children’s Hospital, the morning feels different: quieter, more methodical, and the steady hum of equipment in the background. Dr. Jones steps off the elevator and pauses outside of an office door that’s been left half open. He knocks softly on the door.
“Come on in,” says Dr. Prescott as he sits at his desk, holding a cup of coffee. Next to his computer sits a row of 3D-printed heart stent prototypes lined neatly on a cloth under his desk lamp. He sets down his cup of coffee and grabs the newest model with a pair of tweezers, delicately inspecting the intricate detail of the device.
Dr. Jones steps inside. “I hope I’m not interrupting anything,” he says.
“Not at all,” Dr. Prescott replies, gesturing toward the chair across from his desk. Before closing his computer, he checks his email for an update from the FDA. Not yet.
Dr. Jones sits. “I wanted to update you on Lila. She’s stable, but she isn’t improving. The medications are keeping her level, but we’re not getting any improvement.”
Dr. Prescott nods, not surprised. “I reviewed her test results last night. I had a feeling that was the case.”
“She’s reaching the point where medication isn’t enough.” Dr. Jones says. “The stent you’re developing is exactly what she needs.”
Dr. Prescott leans back slightly in his chair. “We’re close to FDA approval, nearing the end of our Premarket Approval. The reviewers have asked me for one final meeting to confirm the data from our 3D print lab. If they’re satisfied, we could see approval within a couple of weeks.”
Dr. Jones nods slowly. “If approval comes soon, how fast can we move on this?”
Dr. Prescott looks down as he thinks. “We listed St. Edwards Hospital’s 3D Print Lab as a manufacturer on our application. We can print the first units here. Once the approval letter comes in, production can start immediately. There’s no manufacturing delay.”
Dr. Jones rises from his chair. “Alright. This gives us a path forward. Keep me updated on the meeting.”
“I will,” says Dr. Prescott.
After the door closes, the office returns to the quiet humming of 3D printers running in the background. Dr. Prescott returns to the 3D printed stent on his desk, mentally running through the final steps in approval and how he will answer the reviewers’ questions.
“We’re close,” he says to himself, returning to his notes.
* * *
Lila’s room feels heavier than it did last week. She lies tucked under a pink blanket, her eyes half-open, breathing quick and shallow. Her sketchbook is still on the bedside tray, closed, the pencil resting across it untouched since yesterday. She hasn’t had the energy to draw.
Julie sits beside her, one hand resting gently between Lila’s shoulder blades. Her other hand holds the edge of the blanket, gripping it anxiously as she watches her daughter struggle with each breath.
Dr. Jones stands with his arms crossed by the bedside monitor, his tablet catching a glare from the window. He shifts and scrolls through Lila’s morning vitals. The concern is plain on his face. Her heart rate is higher than last week. Her blood pressure, too.
Julie doesn’t look away from Lila. “What happens next?” She asks.
“For now, I’ll adjust her medications again, and we will monitor her closely.” He taps his notes into his tablet. “But I want you to know, we may need to move toward a different option soon.”
Julie nods, reluctantly absorbing this news the same way she has ever since Lila got sick.
He gives Lila a small, steady smile before leaving room 513. Behind him, the soft click fills the room.
Down the hall, Dr. Jones walks past a row of identical rooms, each holding a different world behind it. The overhead lights hum softly, and the faint echo of a rolling supply cart trails from somewhere in the di stance.
His phone vibrates in his coat pocket.
He takes it out of his coat pocket and looks at the screen.
“Call from Dr. Prescott.”
Dr. Jones anxiously presses “Answer.” “The FDA just sent the notice of approval. We have clearance.”
Dr. Jones stops mid-stride. The hallway’s hums narrow. “Is it official?” He asks.
“It’s official,” Dr. Prescott replies. “The approval letter just came through.”
Dr. Jones gives a small nod, already turning toward the elevators. “Alright. I’m on my way up.”
* * *
The elevator doors open onto the research wing with a soft chime. This floor always feels a little removed from the rest of the hospital: quieter, cooler, and more hopeful. Dr. Jones walks down the hall toward Dr. Prescott’s office, the door already cracked open.
Inside, Dr. Prescott sits at his desk with the blinds halfway raised, letting a clean stripe of sunlight fall across the room. The glow catches the edges of the equipment on his shelves and the row of stent prototypes arranged neatly beneath his lamp. On the dual monitors in front of him, cross-sectional images and three-dimensional renderings rotate slowly. Lila’s most recent imaging scans are displayed in precise detail.
He stands when Dr. Jones enters the room. “I pulled her imaging as soon as the notice came in,” Dr. Prescott says, motioning him to come closer. “We’ve already started the patient-matched modeling.”
Dr. Jones steps beside him, eyes flickering back and forth between Lila’s imaging scans and the 3D rendered models. The imaging shows the narrowing in Lila’s artery clearly. Beside it, a digital model of the stent rotates with smooth precision.
“This is the preliminary design,” Dr. Prescott explains. “The software uses the data from her scan to fit the stent’s diameter, curvature, and length. We are going to refine the density this afternoon.”
“How soon until it’s ready to print?” Dr. Jones asks.
“Soon,” Dr. Prescott says. “The FDA approved the hospital’s 3D print lab to manufacture patient-matched units. That’s what we’re waiting on. Once the model is finalized, it can go straight to the printer downstairs.”
He switches to another screen. Photos of the hospital’s 3D printing facility show printers sealed behind glass and technicians in sterile gowns preparing the build trays.
“The process is straightforward now that we’re cleared,” Dr. Prescott continues. “We finalize the model, print the device, we heat treat it, and then it will undergo an inspection.”
Dr. Jones nods, following along with each step. “Assuming everything goes smoothly, we could be ready for the procedure within a few days?”
“That’s right,” Dr. Prescott says. “If the print runs clean, we could have a finished and inspected device by tomorrow afternoon.”
Dr. Jones takes a long look at the final rendering. The delicate curvature of the device is shaped entirely around Lila’s imaging scans. “She and her mother will need to understand the details.”
“They will get them,” Dr. Prescott replies. “I’ll come down this afternoon. I want to make sure they understand what this device is capable of and walk them through the procedure myself.”
Dr. Jones nods. “They’ll appreciate that.”
He leaves the office, each step fading further down the corridor. Dr. Prescott stands for a moment longer, watching the stent slowly rotate. He saves the model and begins organizing the files for the consent discussion with Lila and Julie.
* * *
A gentle knock sounds on the door of Room 513.
Julie looks up from Lila’s bedside as the door opens slowly. Dr. Jones steps in, followed closely by Dr. Prescott and Nurse Ellen, who is also a certified Spanish interpreter. All three enter with calmness and familiarity that follows a team who has worked together for years.
Julie straightens up in her seat, wondering why so many people came to speak with her and Lila. “Is everything okay?” She asks.
Dr. Jones nods and offers a reassuring smile. “Yes. We have some important updates, and we wanted to come together so you wouldn’t have to hear things in bits and pieces.”
Nurse Ellen steps to Julie’s side, giving her a warm, steady smile. “I’m here in case anything needs to be clarified,” she says gently, “We just don’t want any detail to be misunderstood. These conversations can get complicated.”
Julie exhales, comforted by the support. “Thank you.”
Dr. Prescott sets a tablet and a neatly organized folder on the rolling table tray. He meets Julie’s eyes, then Lila’s.
Dr. Jones pulls up a chair and sits so that he is level with them. His voice is soft but steady with a hint of urgency that he cannot disguise.
“The device we discussed,” he begins, “the heart stent. It’s officially been approved.”
Julie and Lila look at each other, wide-eyed.
Dr. Jones continues, “The FDA issued clearance this morning. That means the device can now be used to treat Lila’s condition.”
“And because our hospital’s 3D print lab was included in the submission, we’re authorized to manufacture Lila’s device right here.”
Julie nods slowly, absorbing the words. “So . . . this is really happening?”
“Yes,” Dr. Prescott says. “But before we move forward, there’s a process we need to go through together.”
He opens the folder. Inside are two versions of the informed-consent packet. One in English, and one in Spanish.
“This is the informed-consent documentation,” he explains. “And I want to be very clear about how this works, because it’s important.”
Nurse Ellen stands quietly at Julie’s shoulder, ready if needed.
Dr. Jones continues, “Because Lila is sixteen, she cannot legally give full consent for this procedure. The law requires a parent or legal guardian to make the medical decision on her behalf.”
Julie takes this in with a small, tight nod. “Okay.”
“But,” Dr. Prescott adds, turning slightly toward Lila, “that does not mean Lila is left out. At her age, she participates through what we call assent. We explain everything in a way she can understand, and she tells us if she wants the procedure.”
Dr. Jones nods. “We won’t move forward unless Lila agrees.”
Nurse Ellen watches Julie carefully. “If you want anything repeated or explained more slowly, just ask,” she says kindly.
Julie rests her hand gently on Lila’s. “I understand. Please keep going.”
Dr. Prescott opens his tablet so the screen faces them. “This stent isn’t taken off a shelf,” he says. “It’s created using Lila’s own imaging scans. We take the CT data, build a digital model of the exact shape and size of her narrowed artery, and the printer produces the device layer by layer with medical-grade material. Everything —the design, the materials, and the post-processing —is part of the FDA-approved system.” He pauses, letting the information settle. “It will be printed in our hospital’s 3D print lab downstairs. On-site. For Lila.”
Dr. Prescott taps the tablet screen, bringing up a simplified image of an artery, narrowed on one side and open on the other. “This is what the procedure is designed to fix,” he explains. “The stent is modeled using Lila’s imaging. Because the device is matched to her anatomy, it should fit her artery with far greater accuracy than repurposing an adult stent.”
Lila lifts her eyes from the tablet, her voice quiet but steady.
“So . . . is this like a custom device? Like you’re making it just for me?”
Julie looks toward Dr. Prescott, clearly wondering the same.
Dr. Prescott shakes his head gently. “That’s a great question, Lila. And the answer is no, this isn’t considered a custom device.”
He sits forward slightly, making sure they both understand. “The FDA has a special name for this kind of device: a patient-matched device.”
Julie frowns softly. “What’s the difference?”
Dr. Prescott turns the tablet back toward them and brings up a simplified diagram of the FDA categories.
“A custom device is something hospitals can only use in very rare situations. Those are made when a patient’s needs fall completely outside any normal device design. They bypass the usual approval process, but only in extreme or unusual cases. They’re one-off tools, made when nothing else exists.”
He pauses, then taps to the next image, a digital rendering of Lila’s artery and the stent shaped to fit it.
“This stent belongs to an FDA-approved system. The design, the materials, the printing process, and the sterilization, all of that has been reviewed and cleared. The only thing that changes from patient to patient is the size and the shape of the stent.”
He taps at the screen.
“We stay within an approved range of sizes. But we adjust the exact fit based on the person’s imaging. That’s why it works well for children and teenagers.”
Lila studies the picture. “So it’s . . . mine, but not like some experimental one-of-a-kind thing?”
“Exactly,” Dr. Prescott says with a nod. “It’s yours because it’s shaped for you. But it’s not experimental in the sense of being untested or created outside the rules. We’re personalizing an already-approved design by choosing the best fit for you.”
Julie lets out a slow breath. “That actually makes me feel better.”
“That’s the goal,” Dr. Prescott says, his tone warm but serious.
Dr. Jones gently shifts the tablet so both of them can see a new diagram. “Now let’s talk about what the actual procedure involves,” he says.
“There’s no open surgery. We make a small incision, and from there, we guide the catheter up into the artery and set the stent in place.”
He pauses to let Julie and Lila follow.
“It’s less invasive, the recovery is faster, and we avoid the risks that come with a major operation.”
Nurse Ellen watches Julie closely, stepping in only when Julie hesitates or glances her way for confirmation.
Dr. Jones continues, “There are risks. Bleeding where the catheter goes in. Injury to the vein. Infection. A reaction to the material. And because this device was approved less than 24 hours ago, we don’t have long-term results in teenagers.”
Julie nods, swallowing hard. “I understand.”
Dr. Prescott adds, “But the benefits can be significant: improved blood flow, less strain on her heart, better breathing, and more energy. All of this has been validated through the FDA’s most rigorous premarket approval process.”
Lila sits up in her bed, she listens quietly but attentively, her eyes drifting between the tablet screen and Dr. Jones’s face. Her voice, when she finally speaks, is clear.
“I understand what you’re saying,” she says. “And . . . I think I want to do this. It makes sense.”
Julie turns toward her daughter, then toward the physicians. There is no rush in her movement, no panic, only careful consideration. “You’ve explained everything clearly,” she says to the doctors. “The procedure. The risks. The alternatives. I feel like I understand what this will involve.”
Nurse Ellen stands ready, watching Julie’s expression with practiced ease, but Julie’s nod is confident.
Julie rests her hand on the consent packet. “I’d like to go through this with you,” she says. “Page by page. But based on what we’ve heard, yes, I am prepared to move forward.”
Dr. Prescott nods, appreciative of her careful approach. “That’s exactly how it should be,” he says. “No rush. We’ll go through each part together, and you can ask questions at any point.”
Dr. Jones adds, “And if anything feels unclear, we explain it again. We’re here to make sure you’re fully informed before signing anything.”
Julie gives a small nod. “Thank you,” she says. “That’s important to me.”
Lila shifts slightly beneath her blanket, tired but visibly relieved to have a clear next step laid out. Not driven by fear or desperation, but by understanding.
Room 513 settles into a calm quiet, the kind that comes not from urgency, but from clarity.
A decision made thoughtfully.
A team aligned.
A family choosing the next step with awareness.
* * *
The morning of the procedure arrives with a kind of quiet that feels different from the usual stillness of Room 513. Nurses move a little more purposefully. The halls seem a little calmer. Even the early light through the courtyard window looks softer, as if the hospital itself understands the significance of the day.
Lila is already awake when Julie returns from grabbing a cup of coffee she barely touched. Her sketchbook sits closed on the tray table, its pencil tucked neatly along the spine.
A nurse steps in, offering a gentle smile. “We’re ready whenever you are.”
Julie looks at Lila, tucking the blanket along Lila’s legs.
Lila gives a small nod.
Soon, the transport team arrives with a rolling bed. They introduce themselves with warm, steady voices, and help Lila transition from her room bed to the new one. Everything is calm, practiced, familiar to them. This lends Julie a sense of reassurance.
Dr. Jones meets them in the hallway as they begin to move toward the operating room. He walks beside the bed, offering a warm, grounding smile.
“Everything is ready,” he tells them. “We’ll take good care of her.”
Julie walks alongside until they reach the double doors of the operating room. A nurse turns gently to Julie.
“This is where we take her in,” she says. “You’ll wait right outside. As soon as she’s finished, we’ll bring her back to you.”
Julie leans down and kisses Lila’s forehead. “I’ll be right here,” she whispers.
Lila smiles and gives her a nod.
Julie sits in the nearby waiting area, a quiet alcove with cushioned chairs, a muted television, and a small table covered in magazines no one ever reads. In the background, machines hum softly. Occasionally, a nurse passes through, or a parent stands to stretch their legs. Julie folds her hands in her lap and waits, the minutes dragging on.
After a while, the doors open again.
Dr. Jones walks toward her, pulling off his surgical cap as he approaches. His expression is calm, almost peaceful.
Julie stands before he reaches her.
He gives a small nod.
“The procedure went beautifully,” he says.
* * *
Three months later, Lila runs through the halls of her high school with a group of her friends. It’s the spring art show, and the school is packed. Rows of display boards stand in long lines, each filled with student work. For Lila, it’s a big night: her first time showing her art at school again.
Julie stands beside her daughter’s display, smoothing the edge of her name card. Four of Lila’s pieces hang neatly on the board, painted in soft water colors. Students and parents drift past, many pausing to look.
The doors open, and Dr. Jones and Dr. Prescott step inside. They blend easily into the crowd but walk straight toward Julie with quiet purpose. Dr. Prescott carries a small, neatly wrapped box.
Lila sees them and breaks away from her friends, smiling. “You came!”
“We’re here to celebrate you,” Dr. Jones says.
Prescott hands her the gift. “Open it. We remembered your favorite thing to draw.”
Inside the box is a 3D-printed Wonder Woman figure, simple, the pose steady and strong. Lila’s smile widens.
“No way,” she says softly. “She was the first thing I drew in the hospital!”
“We didn’t forget,” Dr. Jones says.
Lila places the figure at the corner of her display, stepping back to make sure it stands straight. Her friends gather around, admiring it.
Dr. Jones watches her quietly and turns to Julie. “She looks good.”
Julie nods. “She’s herself again.”
Lila straightens the Wonder Woman figure one more time. Standing in front of her artwork, she looks like the small figure she just placed on the display.
Steady.
Healthy.
Strong.
Appendix of Legal Issues
The Food and Drug Administration (FDA) regulates medical devices through a statutory and regulatory framework, which differs from the regulatory framework governing FDA drug approval. This device framework governs risk, premarket review, manufacturing controls, and post market oversight.[1] This applies equally to conventionally manufactured and additively manufactured (3D printed) devices.[2]
- Device Classification and Regulatory Pathways
The FDA categorizes medical devices into three classes— Class I, Class II, and Class III— based on the level of control needed to assure safety and effectiveness for patients.[3] 21 CFR. § 860.3 provides the definitions for each category and the criteria for classifying medical devices.[4]
Class I devices are the lowest-risk category. The FDA requires only general controls, like proper labeling and registration for these devices.[5] A device falls into Class I when these general controls are enough to ensure safety and effectiveness, and the device is not life-supporting or critical in preventing a life-threatening illness.[6]
Class II devices require additional “special controls.”[7] These controls include performance standards, post market surveillance, patient registries, or FDA-issued guidelines.[8] A device is placed in Class II when general controls alone are not enough, but the FDA has enough information to identify what is necessary to ensure safety and effectiveness. Most moderate-risk devices fall within Class II.
Class III devices are the highest-risk category and are defined as those that require premarket approval (PMA).[9] A device is categorized in Class III when it is life-supporting, life-sustaining, crucial to preventing serious health impairment, or poses a potential unreasonable risk of illness or injury.[10] Class III devices must go through PMA because they are the highest-risk devices.[11] PMA is the most demanding type of review the FDA performs, and the manufacturer must receive FDA approval before it can market the device.[12] The application will be approved if it includes sufficient valid scientific evidence showing that the device is safe and effective for its intended use.[13] This evidence comes from detailed scientific and clinical data that FDA examines closely.[14]
Typically, Class I or II devices can go through a different regulatory pathway: the 510(k) pathway. This requires manufacturers to demonstrate that the device is “substantially equivalent” to a legally marketed predicate device.[15] Substantial equivalence depends on whether the device shares the same intended use and similar characteristics to the predicate device.[16] Clinical data is not always required under the 510(k) approval pathway.[17] Devices cleared under this pathway are described as “FDA-cleared,” not approved.[18]
This risk-based framework applies equally to 3D-printed devices.[19] The FDA emphasizes that these guidelines discuss the manufacturing methods used and does not change the device’s classification.[20]
- Additive Manufacturing and Patient Matched Devices
In 2017 the FDA published a guidance document that provides a framework for evaluating 3D-printed devices.[21] This guidance document is not legally binding and only reflects FDA’s current thinking but strongly influences review expectations, much like the FDA’s past guidance in areas such as electronic nicotine delivery systems e-cigarettes.[22]
Patient-matched devices rely on imaging data, like CT scans, and 3D modeling software to generate individualized devices.[23] Additive manufacturing can introduce variability through print orientation, layer thickness, extrusion parameters, material sourcing, and support structures. Accordingly, the FDA expects demonstration of reproducibility, material consistency, and sufficient mechanical strength across the validated range of sizes.[24]
The FDA distinguishes patient-matched devices from custom devices under the Custom Device Exemption.[25] Most additive manufacturing devices will fall within their regular device class under the traditional framework.[26] Patient-matched devices vary within a validated design specification range, meaning individualized measurements must remain within preapproved dimensional and performance limits.[27] These devices remain subject to ordinary PMA or 510(k) requirements.
III. “Manufacturer” Under Regulatory Framework
Federal law defines “manufacturer” broadly to include any person or entity that designs, manufactures, fabricates, assembles, or processes a finished medical device.[28] Because 3D printing and post-processing can constitute device manufacturing, a hospital that performs these activities may be treated as a manufacturer and must meet applicable requirements such as registration and listing under 21 CFR Part 807, Quality System Regulation compliance under 21 CFR Part 820, and medical device reporting.[29]
A hospital that prints devices assumes all of the duties and responsibilities of a traditional manufacturer.[30] However, if a hospital prints devices using a cleared medical device production system (MDPS) and follows its instructions, the MDPS manufacturer is generally responsible for compliance.[31] The FDA requires that a device maintains the same specifications regardless of the manufacturing location, meaning that each printing site must adhere to the same validated, FDA-reviewed process for the device to remain in compliance.[32]
IV. Informed Consent and Assent for Minors
For most patients under eighteen, the law requires a parent or legal guardian who can give informed consent for non-emergency medical care.[33] Minors usually don’t have the legal capacity to fully consent on their own, [34] b ut they are still part of the process. Doctors are expected to explain what’s going on and what the procedure involves in language the child or teenager can understand, then see if the young patient is willing to go ahead. That agreement is called assent.[35] Assent doesn’t replace the consent of a parent or guardian, or make the child the final decision-maker, but it acts as an extra safeguard that ensures minor patients are involved in the decision- making process.[36]
When a parent or child has limited English proficiency, Title VI of the Civil Rights Act requires hospitals and other federally- funded providers to offer meaningful language- accessible services.[37] The Supreme Court treats discrimination based on language the same as discrimination based on national origin.[38] Relying on family members has led to serious misunderstandings and leaves vulnerable patients at a high risk of consenting to procedures they don’t understand.[39] For informed consent to be legally and ethically valid in this setting, parents must get the information in a language they can understand and a qualified interpreter must be involved throughout the discussion to ensure accurate communication.[40] This level of language access is not optional— it is required to receive federal health-care funds.[41]
[1] U.S. Food & Drug Admin., Device Advice: Comprehensive Regulatory Assistance: Overview of Device Regulation, https://www.fda.gov/medical-devices/device-advice-comprehensive-regulatory-assistance/overview-device-regulation. (Feb. 2, 2026).
[2] U.S. Food & Drug Admin., Technical Considerations for Additive Manufactured Medical Devices: Guidance for Industry and Food and Drug Administration Staff, 4 (Dec. 5, 2017).
[3] 21 C.F.R. § 860.3 (Oct. 5, 2021 ).
[4] Id.
[5] Id.
[6] Id.
[7] Id.
[11] U.S. Food & Drug Administration, Premarket Approval (PMA), FDA (May 16, 2019), https://www.fda.gov/medical-devices/premarket-submissions-selecting-and-preparing-correct-submission/premarket-approval-pma.
[15] U.S. Food & Drug Administration, Premarket Notification 510(k), FDA (Aug. 22, 2024), https://www.fda.gov/medical-devices/premarket-submissions-selecting-and-preparing-correct-submission/premarket-notification-510k.
[17] U.S. Food & Drug Administration, Medical Device Safety and the 510(k) Clearance Process, FDA (Sept. 6, 2023), https://www.fda.gov/medical-devices/510k-clearances/medical-device-safety-and-510k-clearance-process.
[19] U.S. Food & Drug Admin., Technical Considerations for Additive Manufactured Medical Devices: Guidance for Industry and Food and Drug Administration Staff , at 4 (Dec. 5, 2017).
[23] Id. at 20.
[24] Id. at 23.
[25] Id. at 8-9.
[29] U.S. Food & Drug Admin., Discussion Paper: 3D Printing Medical Devices at the Point of Care. Page 5 (2021).
[30] Id. at 8-9, 11-12.
[32] Id. at 7.
[33] Aviva L. Katz, Sally A. Webb, & Am. Acad. of Pediatrics Comm. on Bioethics, Informed Consent in Decision-Making in Pediatric Practice, 138 Pediatrics e20161485 (2016).
[37] Alice Hm Chen, Mara K. Youdelman & Jamie Brooks, The Legal Framework for Language Access in Healthcare Settings: Title VI and Beyond, 22 J. Gen. Intern. Med. 362 (2007 ).
[38] Id.; Lau v. Nichols, 414 U.S. 563 (1974).
[39] Chen, Youdelman, & Brooks, supra note 37.